US guidance on CABG vs contemporary PCI in MVD partly driven by ‘misunderstandings, limitations’ of FFR-guided PCI studies
Multivessel disease patients benefit from both CABG and state-of-the-art PCI, expert says, but misinterpretations of limited studies give skewed impressions of CABG superiority
The issue of an optimal revascularization strategy for multivessel disease (MVD) prevailed at COMPLEX PCI 2022 in Seoul, South Korea, with strong claims challenging the major but controversial recommendations from American guidelines that largely endorsed surgery over stenting.
 For MVD patients, Seung-Jung Park, MD, PhD(Asan Medical Center, Seoul, South Korea) advocated an approach that combined invasive revascularization – with either contemporary, state-of-the-art percutaneous coronary intervention (PCI) or conventional coronary artery bypass grafting (CABG) – and guideline-directed medical therapy (GDMT), provided the strategy was tailored to the individual.
For MVD patients, Seung-Jung Park, MD, PhD(Asan Medical Center, Seoul, South Korea) advocated an approach that combined invasive revascularization – with either contemporary, state-of-the-art percutaneous coronary intervention (PCI) or conventional coronary artery bypass grafting (CABG) – and guideline-directed medical therapy (GDMT), provided the strategy was tailored to the individual.
“It’s a field with very limited data, especially PCI-wise,” Park said during the featured lecture session on Nov 25 at the Grand Walkerhill Seoul.

Seung-Jung Park, MD, PhD (Asan Medical Center, Seoul, South Korea) explains the impact of evidence and guideline recommendations for MVD in clinical practice at the COMPLEX PCI 2022 conference held at the Grand Walkerhill Seoul in South Korea on Nov 25.
In 2018, the European Society of Cardiology and European Association for Cardio-Thoracic Surgery (ESC/EACTS) guidelines strongly recommended CABG for triple-vessel disease (3VD) regardless of diabetes status or SYNTAX scores (Class I, A). PCI obtained a Class I (A) recommendation for 3VD patients with low SYNTAX scores (0-22) and a weak Class IIb (A) recommendation for diabetic 3VD patients with low SYNTAX scores. PCI was contraindicated in 3VD patients with intermediate-to-high SYNTAX scores >22 regardless of diabetes status.
In 2021, the American College of Cardiology, American Heart Association and Society of Cardiovascular Angiography and Interventions (ACC/AHA/SCAI) guidelines strongly recommended CABG for multivessel coronary artery disease (MVCAD) patients with anatomy suitable to PCI or CABG and ischemic cardiomyopathy defined as left ventricular ejection fraction (LVEF) <50%). And the strength of CABG recommendations depended on LVEF status: Class 1 recommendations were given for patients with LVEF <35%; Class 2a recommendations were given to patients with LVEF between 35-50%. GDMT was strongly recommended for those unsuitable for CABG, with or without PCI depending on heart team discussions.
Contemporary PCI supported with physiology and imaging is completely different from PCI with first-generation DES.
Endorsement for “any revascularization” with PCI or CABG was relegated to a weak 2b class for 3VD patients and normal LVEF based on the ISCHEMIA (n=5,179) and follow-up ISCHEMIA-EXTEND studies, which showed “nearly identical” outcomes of no survival or ischemic benefits with invasive revascularization compared to GDMT in MVD patients with moderate-to-severe ischemia (>75%).
However, practical application of American guidance was limited in real-world practice, Park said, because few studies supported low LVEF – the major index used to recommend CABG in the 2021 ACC/AHA guidelines – as a guiding factor for revascularization decisions.
“[In the American guidance,] Usefulness of the SYNTAX score was deemed uncertain, and low LVEF (<50%) was the only important index used to recommend CABG,” Park said. “However, the only evidence supporting LVEF as an index are the CASS – from nearly 40 years ago – and STICH studies, which found better outcomes with CABG over medical therapy alone.”
Although CASS (n=780) found no difference in all-cause mortality between CABG and GDMT, there was a statistically non-significant trend favoring surgery over GDMT for triple-vessel patients with stable angina and low LVEF. The STICH study (n=1,212) from 2016 found no differences in 5-year all-cause mortality between patients with stable angina (LVEF <35%) but there was a significant difference between CABG and GDMT groups for 10-year all-cause mortality that favored CABG.
The Class 2b recommendations for either CABG or PCI in MVD patients (>50% LVEF) were based on a handful of studies, including BARI 2D, FREEDOM, SYNTAX and BEST, which reported CABG superiority over PCI in triple-vessel disease.
However, closer analysis of the BEST study, where Park was the leading investigator, revealed no significant between-group differences for 5-year all-cause mortality between PCI and CABG groups, despite the CABG group reporting lower rates of the composite primary endpoint of death, MI or TVR at 5-years.
BEST was the only study to include second-generation DES and report no difference in all-cause mortality between PCI and CABG at 10-years.
The limited number of studies supporting CABG over PCI, along with BEST outcomes showing comparable outcomes, led to minute divisions in language for the equal Class 2b recommendations for CABG (“may be reasonable to improve survival”) and PCI (“usefulness is uncertain”), Park said.
For the diabetic MVD subgroup, the strong recommendation for CABG (Class 1, A) over the Class 2a endorsement for PCI was also rooted in limited and largely misinterpreted studies: “Although BARI 2D, FREEDOM and SYNTAX (3VD Subset) found no benefits with PCI over medical treatment in low-risk patients and better outcomes with CABG over medical treatment in high-risk patients at 5 years, these trials used only first-generation stents (BARI 2D: DES 35% and BMS 56%; FREEDOM: Cypher 49% and TAXUS 41%; SYNTAX: TAXUS 100%).”
“BEST was the only study that had second-generation DES (XIENCE 100%; IVUS 70%) and consequently reported no difference in all-cause mortality between PCI and CABG at 10-years (HR 1.04; 95% CI; 0.65-1.67; P=0.86). Meaning, contemporary PCI supported with physiology and imaging is completely different from PCI with first-generation DES.”
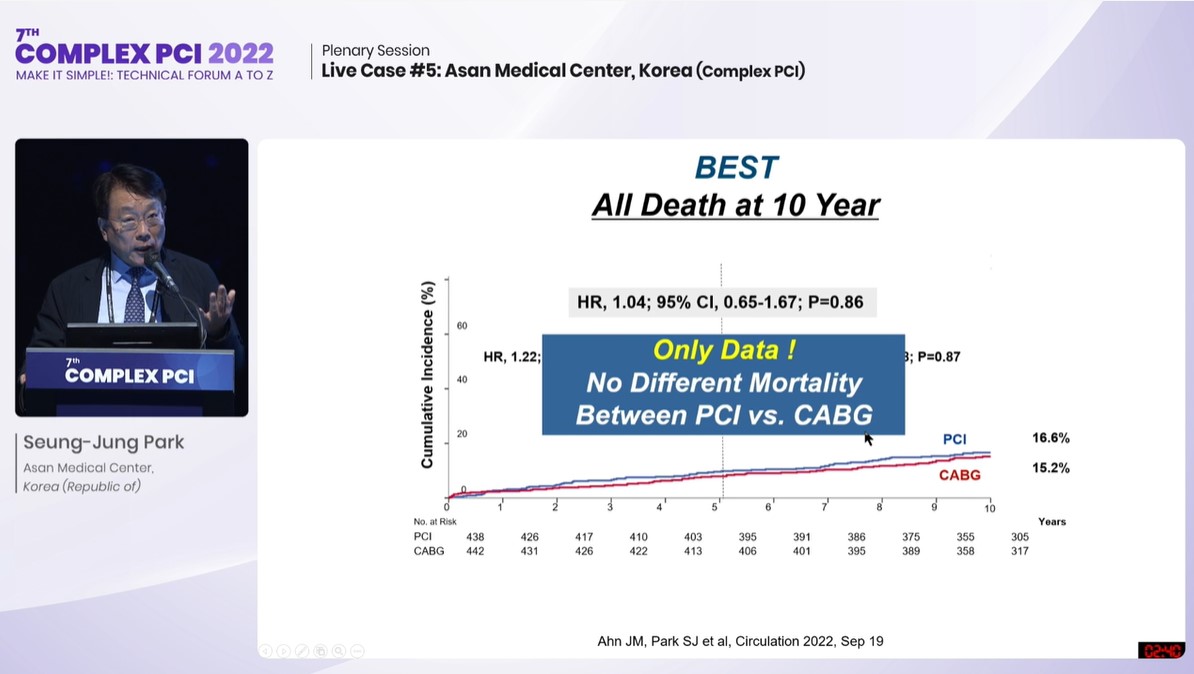
Seung-Jung Park explains results of the extended follow-up outcomes of the BEST trial at COMPLEX PCI 2022.
Demonstrating the impact of physiology and imaging, SYNTAX II (measured lesions: 1,177; IVUS use: 84.1%) found 31% of PCIs were deferred after instantaneous wave-free ratio (iFR) and fractional flow reserve (FFR), and there were no significant differences in 2-year major adverse cardiac and cerebrovascular events (MACCE) between conventional CABG and contemporary PCI for MVD patients (CABG 15.1% vs. PCI 13.2%; HR 0.93; 95% CI; 0.60-1.43; P=0.73).
“The 3 takeaways from SYNTAX II were MACCE reduction with integrated FFR-IVUS use, comparable outcomes between physiology-guided PCI and CABG for 3VD patients, and no association between PCI outcomes and SYNTAX scores,” Park said.
‘Shapeless’ studies regress progress of FFR-guided PCI; complete revasc not stressed enough
Despite FFR improving outcomes for contemporary PCI, poorly designed studies – like the recent randomized RIPCORD-2 study – fueled both misunderstanding and ignorance of FFR and its clinical benefits, which are primarily for intermediate stenosis between 50-80%, Park said.
If 82% of lesions with negative FFR did not influence treatment, what was the point of measuring FFR?
RIPCORD-2 (n=1,100) found systematic FFR assessment did not significantly improve cost or quality-of-life (QoL) over angiography alone; however, the trial included the “unheard of” unicorn of “0-vessel disease” – and the combined 0-vessel and 1-vessel disease patient group accounted for almost 70% of patients: “I mean, FFR was done for people without any disease, so of course, cost increased in the FFR arm.”
The study also bludgeoned the benefits of FFR, which were mostly negative (>0.80) across varying degrees of angiographic severity, by refusing to tailor treatment strategy according to the negative readings for the patient group (mean age 64; male 77%; diabetes 17%; pLAD over 70% <20%).
”75.9% of the angiography arm had one coronary segment targeted for PCI and 70.5% in the angiography plus FFR arm had the same – which was absurd,” Park said. “If 82% of lesions with negative FFR did not influence treatment, what was the point of measuring FFR? The study also compared 2 groups of healthy people so, of course, there was no difference in QoL at 1-year.”
Problems were also cited with the FAME-3 (n=1,500) study for enrolling a 3VD group that would not benefit from FFR guidance. The study reported higher rates of death, MI, stroke or repeat revascularization at 1-year in the combined FFR-guided PCI and OMT group compared to the CABG group (PCI 10.6% vs. CABG 6.9%; HR 1.5; 95% CI; 1.1-2.2; p=0.35 for noninferiority).
“Procedural characteristics showed only 24% in the FFR-guided PCI arm had negative FFR, meaning that 80% of enrolled patients already had tough, ischemic lesions,” Park said. “Considering the major benefits of FFR are localized to intermediate stenosis between 50-80%, FAME-3 dealt with a lesion subset that wouldn’t benefit from FFR.”
Instead of debating “shapeless” studies, he said, the emphasis should be on understanding and raising the completeness of revascularization in contemporary practice.
A patient-level pooled analysis of the SYNTAX, PRECOMBAT and BEST trials, published by Jung-Min Ahn, MD (AMC, Seoul, South Korea) and investigators, found PCI with incomplete revascularization produced the worst rates of all-cause mortality (aHR 1.43; 95% CI; 1.03-2.00; P=0.036) and the composite of death, MI and stroke (aHR 1.48; 95% CI: 1.14-1.92; p=0.003).
Ahn and authors wrote: “For the treatment of left main (LM) or MVCAD, complete revascularization with PCI was associated with a similar long-term survival rate to complete revascularization with CABG, indicating that the ability to achieve complete revascularization should enter the decision algorithm when choosing a revascularization strategy.”
Findings from a systematic review and meta-analysis – which found any revascularization plus GDMT benefited MVD patients compared to medical therapy alone for the outcomes of cardiac death (RR 0.79; 0.67–0.93, P<0.01), spontaneous MI (RR 0.74, 0.64-0.86, P<0.01) and stroke risk (P=0.30) – could further bridge usage of guideline recommendations in clinical practice, Park said.
“Complete ischemic revascularization with DES would be my first choice for all ischemic lesions in MVD with reference vessel diameter (RVD) >2.5 mm, lesion length <50 mm and favorable anatomy for PCI.
“For anatomy unfavorable for PCI, I would consider PCI with DES for the proximal left anterior descending (pLAD) artery with GDMT; when pLAD anatomy or other proximal lesions are unfavorable for PCI (<50% LVEF), then, I would consider CABG.
“CABG would be first for MVD patients with diabetes and low LVEF, but I would also consider multiple DES when anatomy is favorable for PCI.”
Edited by

Do-Yoon Kang, MD
Asan Medical Center, Korea (Republic of)
Written by

