‘More imaging, the better’ to improve clinical outcomes after complex PCI
Do-Yoon Kang, MD explains intracoronary physiology and imaging to optimize complex PCI results based on 2 fundamental questions
 At COMPLEX PCI 2022, interventional cardiologist Do-Yoon Kang, MD(Asan Medical Center, Seoul South Korea) assured there was no “secret skill” for intravascular imaging to achieve optimal imaging criteria during complex PCIs but emphasized that “more is better” to improve outcomes.
At COMPLEX PCI 2022, interventional cardiologist Do-Yoon Kang, MD(Asan Medical Center, Seoul South Korea) assured there was no “secret skill” for intravascular imaging to achieve optimal imaging criteria during complex PCIs but emphasized that “more is better” to improve outcomes.
“Interventionalists have to consider two fundamental questions: is the lesion significant, ischemia-producing and requiring treatment; and is PCI optimized,” Kang said at the conference held at the Grand Walkerhill Seoul in South Korea on Nov 25. “Intracoronary physiology like FFR or iFR helps answer the first question; intracoronary imaging answers the second as a guide for complex PCIs.”

Do-Yoon Kang, MD (Asan Medical Center, Seoul, South Korea) presents strategies for imaging-guided PCI at COMPLEX PCI 2022 in Seoul, South Korea on Nov 25.
FFR and iFR are indices used during invasive coronary angiography to assess the functional significance of coronary stenosis that could cause myocardial ischemia, measured by passing high-fidelity pressure wires distal to the coronary stenosis.
Although both angiography and intravascular imaging can assess lesion significance during complex PCI, the use of angiographic images – a luminographic technique – can be limited for eccentric lesions and diffusely diseased lesions. Contemporary intracoronary imaging modalities like intravascular ultrasound (IVUS) and optical coherence tomography (OCT), Kang explained, help visualize the “real” vessel by providing precise measurements for key factors such as the vessel, lumen diameters and lesion morphology.
Although IVUS and OCT overcomes some angiographic limitations – becoming an indispensable tool for contemporary complex PCIs – their relatively recent inclusion into the cardiologists’ armamentarium has limited standardization in clinical practice, leading to heterogeneity usage across countries, centers and operators.
There’s no secret, master skill for imaging. My advice? Just do it for complex PCIs.
Based on accumulated data, the 2021 American College of Cardiology and American Heart Association (ACC/AHA) guidelines on intracoronary imaging gave a Class of Recommendation (COR) IIa (Level of Evidence, LOE: B) recommendation for IVUS as useful procedural guidance to reduce ischemic events in patients undergoing PCI, particularly in LM or complex coronary artery stenting. OCT was recommended as a reasonable procedure-guiding alternative to IVUS in patients undergoing PCI (COR IIa; B), excluding patients with ostial LM disease. For patients with stent failure, both IVUS or OCT were deemed reasonable to determine the mechanism of stent failure (COR IIa; C).
Despite the relatively weak recommendations and noted underutilization of intravascular imaging in clinical practice, both randomized and non-randomized studies have demonstrated their usefulness in improving PCI outcomes, Kang said, such as the prospective multicenter non-randomized all-comer ADAPT-DES study where IVUS use (39%) prompted changes in strategy for 74%, aiding the selection of larger-diameter devices, longer stents, higher inflation pressures and additional post-dilation.
Large randomized controlled trials (RCTs), like IVUS-XPL and the all-comer ULTIMAT, also found IVUS halved rates of major endpoints compared to angiography-guidance: the former found IVUS-guidance was associated with significantly lower 1-year major adverse cardiac events (MACE) rates over angiography-guidance (2.9% vs. 5.8%; HR 0.48; 95% CI; 0.28-0.83, P=0.007); while the latter showed lower 1-year target-vessel failure (TVF) rates with IVUS-guidance compared to angio-guidance (2.9% vs. 5.4%; HR 0.53; 95% CI; 0.31-0.90; P=0.019).
Bigger balloons and bigger stents make bigger lumen areas, which produce better clinical outcomes.
IVUS-guidance also lowered composite outcome rates (cardiac death, MI) for chronic total occlusion (CTO)-PCIs compared to angio-guidance in the randomized CTO-IVUS study (n=402) by Byeong-Keuk Kim, MD, PhD (Severance Cardiovascular Hospital, Seoul, South Korea) and South Korean investigators (2.6% vs. 7.1%; HR 0.35; 95% CI; 0.13-0.97; P=0.035); and for left main (LM) disease in the MAIN-COMPARE Registry by Do-Yoon Kang, MD and AMC investigators, which found the IVUS-guided group had lower rates of 10-year all-cause mortality (IVUS 16.4% vs. 31.0%; HR 0.54; 0.35-0.65; P<0.001) and the composite of all-cause death, Q-wave MI or stroke (IVUS 19.2% vs. angio 32.9%; HR 0.57; 0.39-0.69; P<0.001) compared to angio-guidance.
Finally, IVUS-guidance reported lower rates of the composite outcome (CVD mortality, MI, TLR and stent thrombosis) compared to angio-guidance in an updated meta-analysis that included 10 RCTs(n=5160) by Islam Y. Elgendy (University of Florida, Gainesville, USA) and colleagues who suggested the findings “should encourage operators to use IVUS more often.”
Role of intravascular guidance in PCI? ‘Optimize acute stent results’
Addressing the ambiguity of an optimal real-world strategy for intravascular imaging, Kang noted that its role in PCI guidance is to “optimize acute stent results,” including its use to:
- Assist guide wiring in complex bifurcation or CTO lesions
- Assess plaque characteristics and perform adequate pre-modification
- Confirm results of plaque modification in calcified lesions
- Evaluate calcium (angle, thickness, length) with imaging-predicted stent expansion to help decision-making for atherectomy or other devices.
- Select clean landing zones (stent length) with larger stents (lumen/vessel size)
- Select larger non-compliance (NC) balloons for post-dilation
- Post-stent surveillance for stent apposition-, expansion- and edge-related problems (dissections, hematoma)
“IVUS helps identify an optimal balloon and stent size along with an optimal length for the landing zone,” he explained. “For edge restenosis, the best cut-off is around plaque burden <55%> - according to a study by Soo-Jin Kang, MD, PhD and AMC investigators - which means landing zones with less plaque burden leads to better outcomes.”
Interventionalists have to consider two fundamental questions: is the lesion significant, ischemia-producing and requiring treatment; and is PCI optimized.
Effective stent area also makes a difference, Kang said, according to a study published by Hae-Geun Song, MD and AMC investigators that found smaller stent area predicted angiographic restenosis for PCIs with both first- and second-generation drug-eluting stents (DES).
“Effective stent area greater than 5.0 ㎟ was associated with lower TLR rates (<2%),” Kang said. “Bigger stent areas are better for LM bifurcation PCIs – and intravascular imaging makes that possible.”
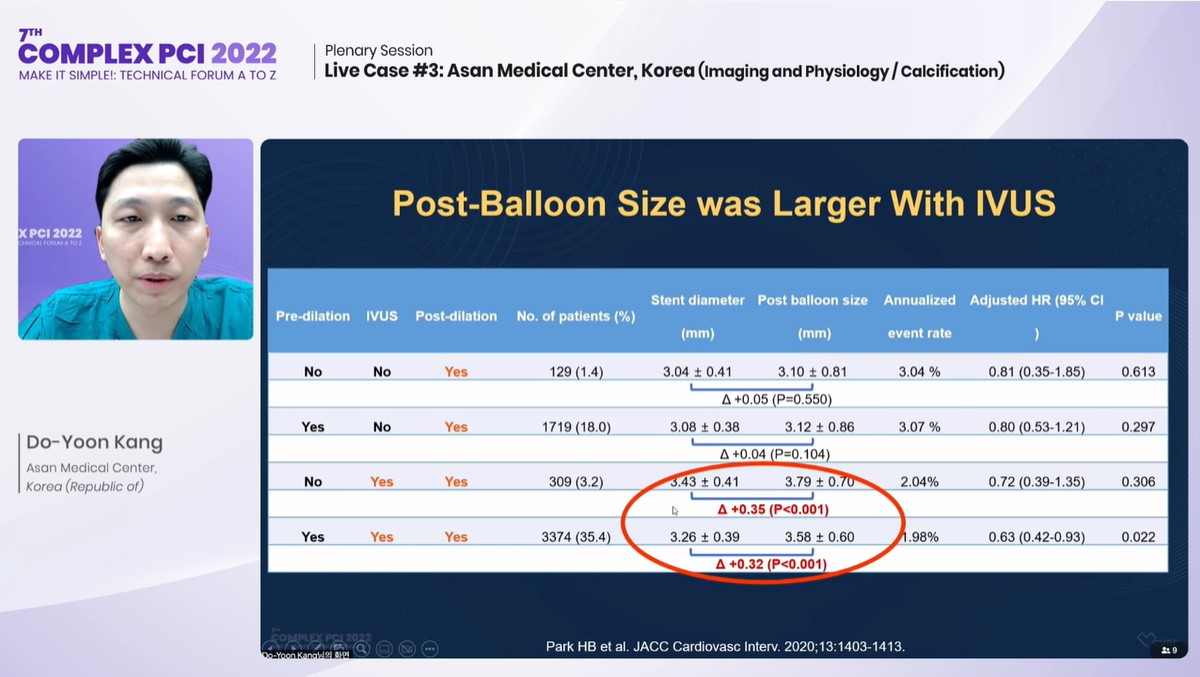
Selecting larger-sized NC balloons for post-dilation is important, as evidenced by the recent IRIS-DES Registry by Hanbit Park, MD, Jung-Min Ahn, MD and AMC investigators that examined rates of cardiac death, target-vessel MI and TVR in 9,525 patients with single, complex coronary lesions (LM, bifurcation, diffuse disease >30 mm, severe calcification and in-stent restenosis) who underwent imaging-guided PCI (iPSP) or PCI without imaging guidance (no iPSP).
Results showed better clinical outcomes with imaging-guided complex PCIs over no imaging guidance. Imaging-guided PCI also led to more stents (iPSP 1.41 vs. no iPSP 1.35; p<0.001), longer stent length (37 mm vs. 33 mm; p<0.001), bigger stent diameter (3.3 mm vs. 3.2 mm; p<0.001) and bigger final balloon size (3.6 vs. 3.4; p<0.001).
Post-dilation (HR 0.79; 0.67-0.94; 0.006) was the most significant event predictor among the major 3 components of pre-dilation (HR 0.89; 0.69-1.15; p=0.374), stent-sizing (HR 0.79; 0.67-0.93; P=0.004) and post-dilation. Larger balloon sizes were used during post-dilation with IVUS (absolute difference 0.3 mm).
Do-Yoon Kang presents findings from the IRIS-DES registry analysis at COMPLEX PCI 2022.
“Small details make a big difference for long-term clinical outcomes,” Kang said. “Intravascular imaging enables safe implantation of bigger stents with higher pressure post-dilation since we know the vessel size.”
A recent patient-level pooled analysis, co-authored by Jun-Jie Zhang (Nanjing Medical University, Nanjing, China) and Severance Hospital’s Sung-Jin Hong, MD and Myeong-Ki Hong, MD, PhD (Seoul South Korea) also found patients with long lesions (≥28 mm) without optimal stent expansion fared worse than those with optimal stent expansion for the primary endpoint of cardiac death, MI or stent thrombosis at 3-years.
The study merged the randomized IVUS-XPL and ULTIMATE studies – two databases with long lesions – and found about half of the patients did not meet the optimal stent expansion criteria after applying various definitions of stent optimization criteria, including minimal stent area (MSA) > 5.5 mm2 (optimal stent expansion: 51.4% vs. no optimal stent expansion 48.6%) and MSA >5.0 mm2 (optimal stent expansion: 65.6% vs. no optimal stent expansion 34.4%).
Small details make a big difference for long-term clinical outcomes.
Stratified by optimization criteria, results showed that the 3-year primary endpoint rates were significantly higher without optimal stent expansion for IVUS-defined optimal stent expansion of MSA >5.5 mm2 (HR 0.21; 0.06-0.75; P=0.008); MSA >5.0 mm2 (HR 0.24; 0.09-0.68; P=0.003); and MSA-distal reference lumen area >90% (HR 0.32; 0.12-0.88; P=0.019).
IVUS-guidance plus post-dilation produced significantly better clinical outcomes, with study results indicating larger minimal lumen area (MLA) in patients who received post-dilation with IVUS (p<0.001) compared to those who received angiography with post-dilation (p=0.473) or angiography without post-dilation (p=0.046);
“Bigger balloons and bigger stents make bigger lumen areas, which make better clinical outcomes,” Kang said. “For angio-guided post-dilation – where we only see the lumen – operators often select stent-to-reference vessel diameter ratios between 1.0 and 1.1. For IVUS-guided post-dilation, we can select stent-to-reference vessel diameter ratios between 1.1 and 1.3.”
Intracoronary imaging strategies can also optimize post-stenting surveillance, he added: “You can visualize the result of stenting and identify problems related to stent apposition, expansion and edge dissections and hematoma. Overall, there’s no secret, master skill for imaging so my advice is just use intravascular imaging for complex PCI and do your best to achieve optimal imaging criteria.”
Edited by

Do-Yoon Kang, MD
Asan Medical Center, Korea (Republic of)
Written by

