CASE20220822_001
Chronic Total Occlusion with In-stent Restenosis and Graft Occlusion after Coronary Artery Bypass Grafting in a 52 year-old Filipino male: a Successful Management
By , ,
Presenter
Thessa Mae Ravena Ecarma-Villareal
Authors
1, 1, 1
Affiliation
, Philippines1
Complex PCI - Chronic Total Occlusion
Chronic Total Occlusion with In-stent Restenosis and Graft Occlusion after Coronary Artery Bypass Grafting in a 52 year-old Filipino male: a Successful Management
1, 1, 1
, Philippines1
Clinical Information
Patient initials or Identifier Number
JG
Relevant Clinical History and Physical Exam
Case of a 52 year-old Filipino male, hypertensive, who had ACS-STEMI in 2009 and underwent PCI of the LAD and LCX. In 2014, there was recurrence of chest pain with new-onset AF. Repeat coronary angiogram showed in-stent restenosis in the LAD and totally occluded proximal RCA thus he underwent bypass with two grafts (LIMA to LAD and SVG to RPDA). In the interim, persistent dyspnea was noted; angiogram showed graft occlusion, type IV in-stent restenosis in the LAD and CTO of the proximal RCA.
Relevant Test Results Prior to Catheterization
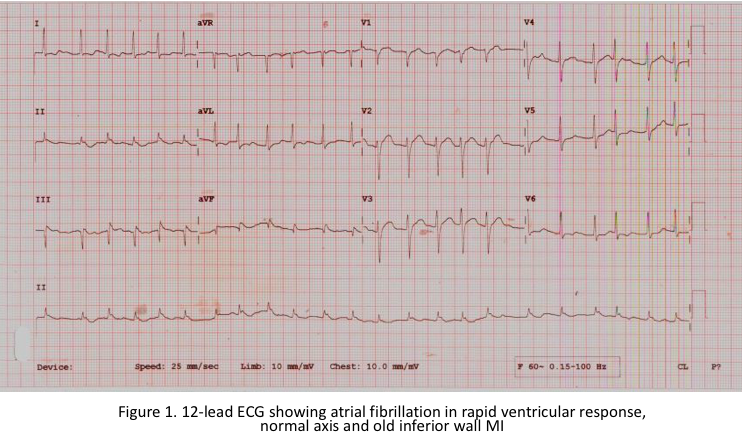
12-lead ECG showed atrial fibrillation in rapid ventricular response, old inferior wall myocardial infarction. Echocardiography showed concentric LV hypertrophy with hypokinesia of the anterolateral and inferior left ventricular free wall from base to apex. The ejection fraction is 53%, with adequate RV contractility. Myocardial perfusion imaging showed moderate to severe stress-induced myocardial ischemia in the basal to apical anterior and inferior segments, all myocardial segments are viable.

Relevant Catheterization Findings
The left main has luminal irregularities, trifurcating into the LAD, ramus intermedius and LCX. The LAD has 95% ostio-proximal segment stenosis with total occlusion of the proximal segment of the previously deployed stent with the distal segments visualized via collaterals. The LIMA-LAD graft is patent up to the mid-segment only.The LCX is non-dominant with patent stent and 40% distal segment stenosis; RCA is totally occluded at the proximal segment with distal segments visualized via collaterals.
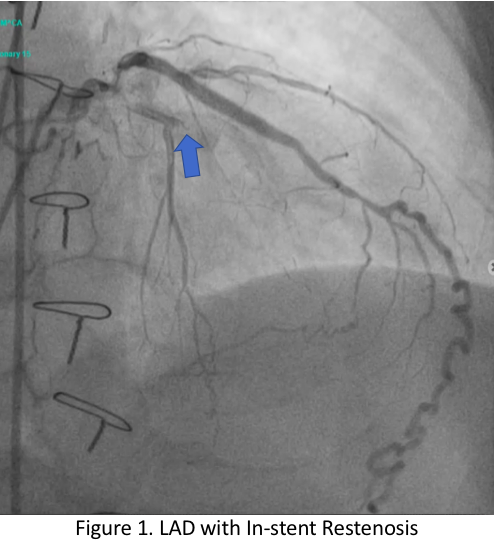
 JG LAD Angiogram.mov
JG LAD Angiogram.mov
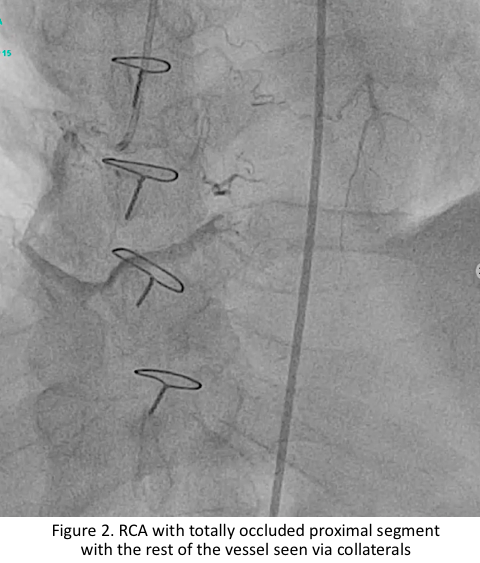
 JG RCA Angiogram.mov
JG RCA Angiogram.mov
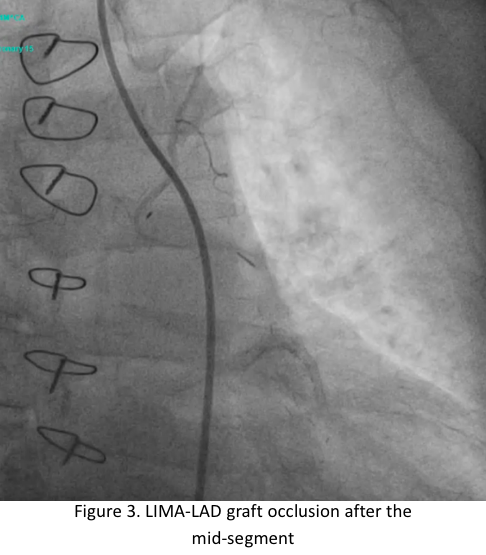
 JG Graft Angiogram.mov
JG Graft Angiogram.mov



Interventional Management
Procedural Step
Patient underwent PCI of the LAD and the RCA. Dual injection was performed at the start of the procedure to visualize the distal vessel. Antegrade wire escalation technique was used to cross the CTO lesion. IVUS imaging was performed by slow pullback using manual technique and confirmed type IV in-stent restenosis in the LAD.
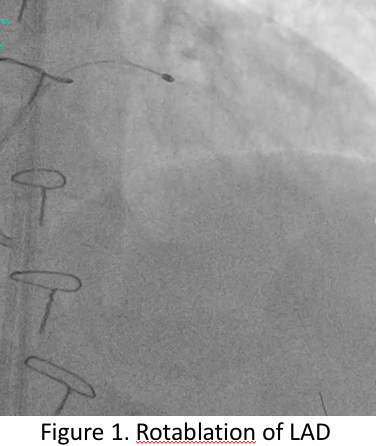
 Video A Rotablation of LAD.mov
Video A Rotablation of LAD.mov
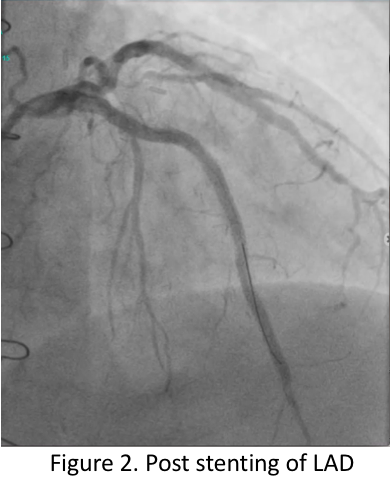
 Video B Post LAD Stenting.mov
Video B Post LAD Stenting.mov
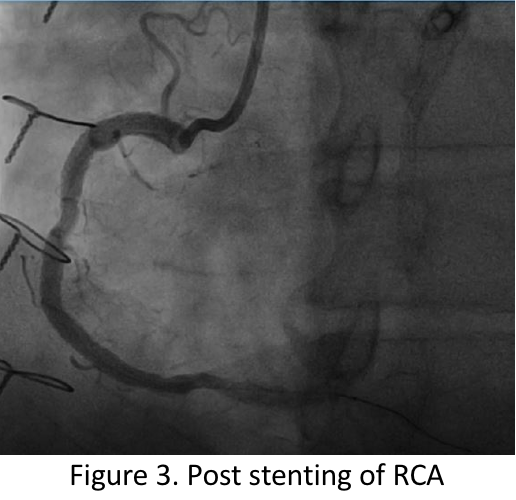
 Video C Post RCA Stenting.mov
Video C Post RCA Stenting.mov



Case Summary
Repeat revascularization following ISR is a complex procedure; early detection and timely intervention are important. Even in the absence of usual risk factors such as diabetes, ISR should still be considered in patients experiencing reappearance of symptoms. Coronary angiogram identifies ISR but IVUS may elucidate the mechanism and guide stent implantation. In patients with ISR and complex multivessel disease such as in our patient, CABG is preferred. However, with graft occlusion post CABG, ISR persistence, and CTO of the native vessels, repeat PCI with DES, IVUS and rotablation were key to this successful revascularization, prevention of mortality and overall improved functional capacity.
